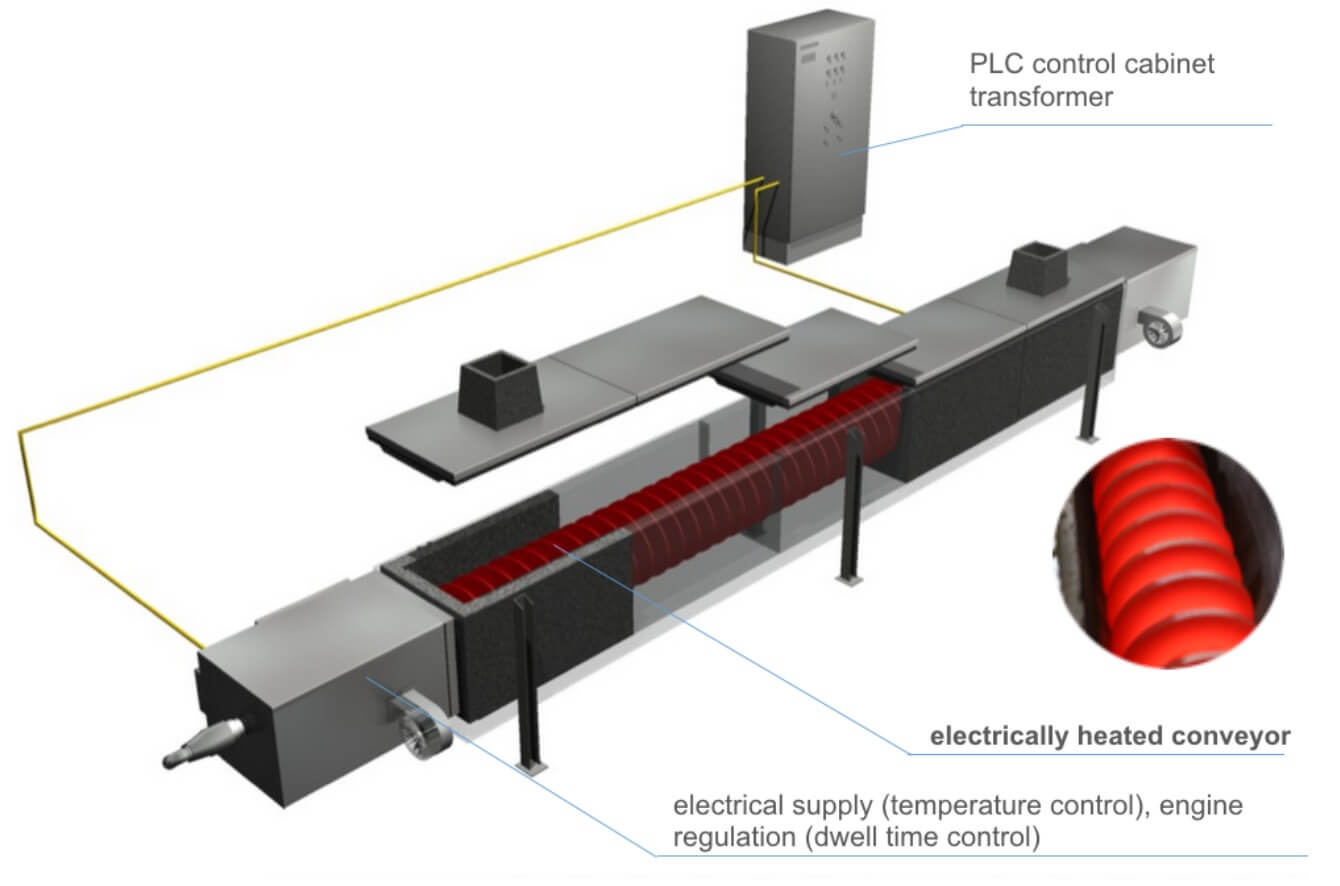
Calcination is a thermal treatment process most commonly applied to inorganic products. In its most conventional meaning, the term “calcination” is widely used in industrial thermal processing terminology to describe the processes of burning the lime or converting the iron ores into oxides. In the modern technologies, the definition has been widened and calcination is often used to describe a variety of process of decomposition through heating in controlled atmospheres: both oxidizing and inert, performed to achieve a certain degree of material transformation, without its reaching its melting or fusing point.
Today, most common examples of commercial calcination processes using high temperatures include operations of chalk or limestone conversion to lime, dehydration of minerals, activation of carbon, catalyst regeneration, driving off the carbon dioxide, as well as treatment eliminating the undesired compounds like volatiles or organics.